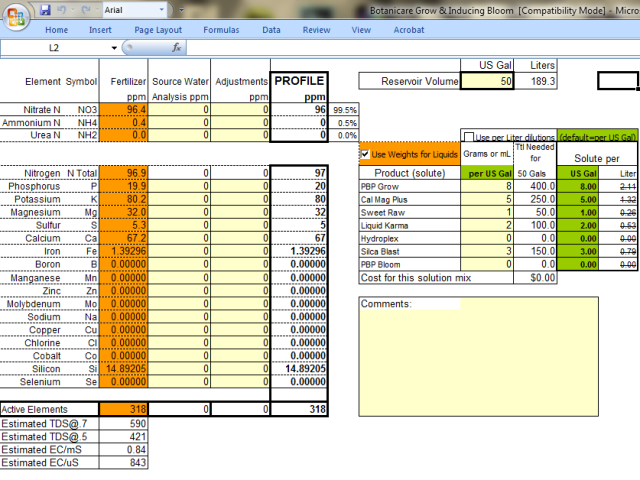
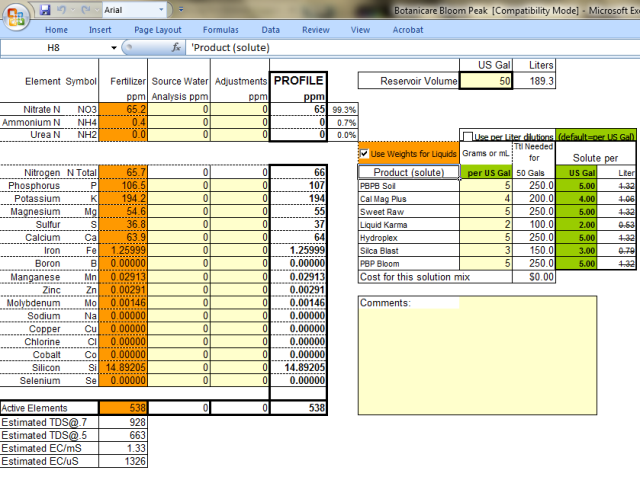
Ok so from my perspective, using the ratios you've provided, you are feeding as follows. I could be wrong, but this is just using the math skills that I know and love based on guaranteed analysis of liquids.
Veg NPK is exactly 2-1-3
Transition NPK is roughly 1-1-2
Flower NPK is exactly 3-4-5
If you are interested in how I came to these numbers,
it's actually pretty simple you just have to multiply in each part by the npk and add it up. so for veg
3 parts grow (3 x 2-1-6) = 6-3-18 + 2 parts micro (2 x 5-0-1) = 10-0-2 + 1 part bloom 0-5-4 = 16-8-24 which reduces to 2-1-3.
I could explain it further also.. would love it someone else could chime in with their thoughts
I believe that your calculations are correct. Potassium is important for plant growth in both vegetative growth and flower.
Potassium - Wikipedia, the free encyclopedia
"Fertilizer
Potassium and magnesium sulfate fertilizer
Potassium ions are an essential component of plant nutrition and are found in most soil types.[7] They are used as a fertilizer in agriculture, horticulture, and hydroponic culture in the form of chloride (KCl), sulfate (K
2SO
4), or nitrate (KNO
3). Agricultural fertilizers consume 95% of global potassium chemical production, and about 90% of this potassium is supplied as KCl.[7] The potassium content of most plants range from 0.5% to 2% of the harvested weight of crops, conventionally expressed as amount of K
2O. Modern high-yield agriculture depends upon fertilizers to replace the potassium lost at harvest. Most agricultural fertilizers contain potassium chloride, while potassium sulfate is used for chloride-sensitive crops or crops needing higher sulfur content. The sulfate is produced mostly by decomposition of the complex minerals kainite (MgSO4·KCl·3H2O) and langbeinite (MgSO4·K2SO4). Only a very few fertilizers contain potassium nitrate. In 2005, about 93% of world potassium production was consumed by the fertilizer industry."
Higher phosphorous is required in flower.
Phosphorus - Wikipedia, the free encyclopedia
"Fertiliser
Main article: Fertiliser
The dominant application of phosphorus is in fertilisers, which provides phosphate as required for all life and is often a limiting nutrient for crops. Phosphorus, being an essential plant nutrient, finds its major use as a constituent of fertilisers for agriculture and farm production in the form of concentrated phosphoric acids, which can consist of 70% to 75% P2O5. Global demand for fertilisers led to large increase in phosphate (PO43−) production in the second half of the 20th century. Due to the essential nature of phosphorus to living organisms, the low solubility of natural phosphorus-containing compounds, and the slow natural cycle of phosphorus, the agricultural industry relies on fertilisers that contain phosphate. A major form of these fertilisers is superphosphate of lime, a mixture of two salts, calcium dihydrogen phosphate Ca(H2PO4)2 and calcium sulfate dihydrate CaSO4·2H2O, produced by the reaction of sulfuric acid and water with calcium phosphate.
So large is the need to process phosphate minerals with sulfuric acid to obtain fertiliser, that this use is the major global industrial use for important industrial chemical sulfuric acid (the production of which, in turn, is the largest industrial use of the element sulfur).
Widely used compounds Use
Ca(H2PO4)2·H2O Baking powder and fertilizers
CaHPO4·2H2O Animal food additive, toothpowder
H3PO4 Manufacture of phosphate fertilizers
PCl3 Manufacture of POCl3 and pesticides
POCl3 Manufacture of plasticizer
P4S10 Manufacturing of additives and pesticides
Na5P3O10 Detergents"