Hello All,
Been lurking for a while and soaking up as much as possible.
Finally ran into my first issue that I can't beat.
Stats:
Growing bagseed (Possibly SAGE)
Fox Farm Grow - Ocean Forest Soil, Big Bloom, Tiger Bloom.
4tsp Tiger Bloom, 8tsp Big Bloom per Gallon, 2-3 times per week.
(I know this seems like a lot, but i'm seeing deficiencies and not burn..Please read on)
Flush with FF Sledgehammer every 4 weeks.
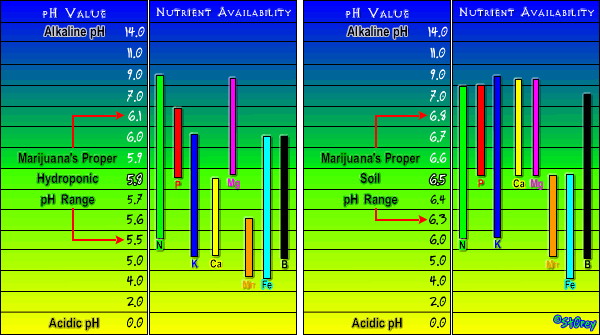
Water Ph into soil is 6.5, run off is roughly 5.8-6.0
Flowering under a 600w Diamond LED from AdvancedLED.
History:
I will post pics of three different plants, all in flower, all clones from the same mother.
No issues while cloning or vegging, all healthy and vibrant.
Clones vegged for roughly 10 days before moving into flower.
Upon moving into flower, transplanted to 2 gallon pots.
First 2 weeks of flower go off without a hitch.
by the second week I begin to notice a slight yellowing between the veins of lower leaves.
2nd week Exmple 1:

2nd week Example 2:

By the 4th week of flower I get red/purple stems.
I begin to see definitive signs of a Phosphorus def.
(Small brown necrotic spots on the edges of the leaves slowly moving inward)
I have kicked up the Phosphorus and no improvement.
4th week Example 1:

4th week Example 2:

By the 5th week I'm basically losing most of my large fan leaves.
All symptoms above begin to creep into all smaller leaves from the middle of the plant and down.
Runoff Ph is slightly lower but never below 5.5.
I have upped my dose of Big Bloom to 10tsp with no effect.
5th week Example 1:

5th week Example 2:

Last for you I have the sad and unfortunate first clone that was flowered.
She is just destroyed by the slow progression of whatever evil is afflicting these poor girls! Please help!
Sad:

Sad 2:

-Evlded
Been lurking for a while and soaking up as much as possible.
Finally ran into my first issue that I can't beat.
Stats:
Growing bagseed (Possibly SAGE)
Fox Farm Grow - Ocean Forest Soil, Big Bloom, Tiger Bloom.
4tsp Tiger Bloom, 8tsp Big Bloom per Gallon, 2-3 times per week.
(I know this seems like a lot, but i'm seeing deficiencies and not burn..Please read on)
Flush with FF Sledgehammer every 4 weeks.
Water Ph into soil is 6.5, run off is roughly 5.8-6.0
Flowering under a 600w Diamond LED from AdvancedLED.
History:
I will post pics of three different plants, all in flower, all clones from the same mother.
No issues while cloning or vegging, all healthy and vibrant.
Clones vegged for roughly 10 days before moving into flower.
Upon moving into flower, transplanted to 2 gallon pots.
First 2 weeks of flower go off without a hitch.
by the second week I begin to notice a slight yellowing between the veins of lower leaves.
2nd week Exmple 1:

2nd week Example 2:

By the 4th week of flower I get red/purple stems.
I begin to see definitive signs of a Phosphorus def.
(Small brown necrotic spots on the edges of the leaves slowly moving inward)
I have kicked up the Phosphorus and no improvement.
4th week Example 1:

4th week Example 2:

By the 5th week I'm basically losing most of my large fan leaves.
All symptoms above begin to creep into all smaller leaves from the middle of the plant and down.
Runoff Ph is slightly lower but never below 5.5.
I have upped my dose of Big Bloom to 10tsp with no effect.
5th week Example 1:

5th week Example 2:

Last for you I have the sad and unfortunate first clone that was flowered.
She is just destroyed by the slow progression of whatever evil is afflicting these poor girls! Please help!
Sad:

Sad 2:

-Evlded